Press release
First in the world Sungen Biomedical obtains FDA's clinical trial approval of IND SGC001 as an emergency use antibody drug for AMI
On May 23rd, 2024, Eastern Standard Time, US Food and Drug Administration (FDA) approved the clinical trial application for the world's first AMI antibody drug, SGC001, as an investigational new drug (IND). The drug is developed by Beijing Sungen Biomedical Technology Co., Ltd. (Sungen Biomedical), an innovative biopharmaceutical company incubated by Beijing Hotgen Biotech Co., Ltd. (SH.688068,www.hotgen.com.cn). This marks a breakthrough in innovative drug development of Sungen Biomedical and the overall biopharmaceutical industry.
SGC001 is a monoclonal antibody indicated for emergency treatment of acute myocardial infarction (AMI). The drug is developed by Professor Zhiwei Sun's team from Sungen Biomedical, in collaboration with Professor Jie Du's team from Beijing Institute of Heart, Lung and Blood Vessel Diseases, Capital Medical University. AMI is a condition characterized by death of myocardia due to acute occlusion of coronary arteries, resulting in high morbidity, mortality, disability rate, and various serious complications.
It is estimated that there are 2.5 million deaths from heart attacks annually in China. While millions of new myocardial infarction patients are diagnosed yearly, this group are getting significantly younger. A prospective AMI study PEACE (Patient-centered Evaluative Assessment of Cardiac Events) in 2020 suggests that the number of AMI patients in China could reach 23 million by 2030.
Based on incidence data from the ARIC (Atherosclerosis Risk In Communities) study of NHLBI (National Heart Lung and Blood Institute), AHA (American Heart Association) indicates that approximately every 40 seconds, one American will have an MI. The 2005 to 2014 ARIC study estimates that there are 605,000 new attacks and 200,000 recurrent attacks each year in the US. And 170,000 of these are silent, without significant symptoms.
Till now, there is no antibody therapy for AMI approved for clinical or commercial stage. The successful clinical trial application in the United States is an important milestone of SGC001 and a huge step towards the internationalization of innovative drugs developed by Sungen Biomedical. Preclinical pharmacodynamic and toxicological studies reveales that SGC001 has obvious therapeutic effects on heart failure and pathological remodeling of the heart after AMI, with significantly decreased mortality rate, reduced infarction size, improved cardiac functions, good efficacy and a reliable therapeutic window. SGC001 has the potential to become a first-in-class drug, providing safer and more effective therapies for AMI patients worldwide.
Contact us: bd@sungen.net.cn [mailto:bd@sungen.net.cn]
Media Contact
Company Name: Beijing Sungen Biomedical Technology Co., Ltd
Contact Person: Honggang Zhang
Email: Send Email [http://www.universalpressrelease.com/?pr=first-in-the-world-sungen-biomedical-obtains-fdas-clinical-trial-approval-of-ind-sgc001-as-an-emergency-use-antibody-drug-for-ami]
Country: China
Website: http://www.hotgen.com.cn
SGC001 is a monoclonal antibody indicated for emergency treatment of acute myocardial infarction (AMI). The drug is developed by Professor Zhiwei Sun's team from Sungen Biomedical, in collaboration with Professor Jie Du's team from Beijing Institute of Heart, Lung and Blood Vessel Diseases, Capital Medical University. AMI is a condition characterized by death of myocardia due to acute occlusion of coronary arteries, resulting in high morbidity, mortality, disability rate, and various serious complications.
It is estimated that there are 2.5 million deaths from heart attacks annually in China. While millions of new myocardial infarction patients are diagnosed yearly, this group are getting significantly younger. A prospective AMI study PEACE (Patient-centered Evaluative Assessment of Cardiac Events) in 2020 suggests that the number of AMI patients in China could reach 23 million by 2030.
Based on incidence data from the ARIC (Atherosclerosis Risk In Communities) study of NHLBI (National Heart Lung and Blood Institute), AHA (American Heart Association) indicates that approximately every 40 seconds, one American will have an MI. The 2005 to 2014 ARIC study estimates that there are 605,000 new attacks and 200,000 recurrent attacks each year in the US. And 170,000 of these are silent, without significant symptoms.
Till now, there is no antibody therapy for AMI approved for clinical or commercial stage. The successful clinical trial application in the United States is an important milestone of SGC001 and a huge step towards the internationalization of innovative drugs developed by Sungen Biomedical. Preclinical pharmacodynamic and toxicological studies reveales that SGC001 has obvious therapeutic effects on heart failure and pathological remodeling of the heart after AMI, with significantly decreased mortality rate, reduced infarction size, improved cardiac functions, good efficacy and a reliable therapeutic window. SGC001 has the potential to become a first-in-class drug, providing safer and more effective therapies for AMI patients worldwide.
Contact us: bd@sungen.net.cn [mailto:bd@sungen.net.cn]
Media Contact
Company Name: Beijing Sungen Biomedical Technology Co., Ltd
Contact Person: Honggang Zhang
Email: Send Email [http://www.universalpressrelease.com/?pr=first-in-the-world-sungen-biomedical-obtains-fdas-clinical-trial-approval-of-ind-sgc001-as-an-emergency-use-antibody-drug-for-ami]
Country: China
Website: http://www.hotgen.com.cn
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage
to this press release on woodPRI. woodPRI disclaims liability for any content contained in
this release.
Recommend

/newsMicroencapsulation Market Deep Analysis on Key Players - Dow Corning, Encapsys, Syngenta Crop Protection, Evonik Industries, 3M and Bayer
Market Study Report Adds Global Microencapsulation Market Size, Status and Forecast 2024 added to its database. The report provides key statistics on the current state of the industry and other analytical data to understand the market.
Extensive research is required for choosing the appropriate cor...

/newsGermany Airbag Market Size 2023: Global Share, Industry And Report Analysis By 2030 | Hyundai Mobis Co., Ltd. Key Safety Systems, Inc. Robert Bosch GmbH
Germany airbag market is expected to grow at a CAGR of around 6% during the forecast period. Germany Airbag Market research report refers to gathering and analyzing significant market data serve as best medium for various industry players to launch novel product or service. It is vital for key firms...
/newsSecurities Brokerages And Stock Exchanges Market Outlook 2021: Big Things are Happening
A new intelligence report released by HTF MI with title "Global Securities Brokerages And Stock Exchanges Market Survey & Outlook" is designed covering micro level of analysis by Insurers and key business segments, offerings and sales channels. The Global Securities Brokerages And Stock Exchange...

/newsRenewable Chemicals Market Emerging Trends and Competitive Landscape Forecast to 2028
The renewable chemicals market was valued at US$ 80,566.30 million in 2021 and is projected to reach US$ 1,76,750.76 million by 2028 it is expected to grow at a CAGR of 11.9% from 2021 to 2028. The research report focuses on the current market trends, opportunities, future potential of the market, a...
/newsHow Coronavirus is Impacting Cold Brew Coffee, Global Market Volume Analysis, Size, Share and Key Trends 2020-2026
"Market Latest Research Report 2020:
Los Angles United States, February 2020: The Cold Brew Coffee market has been garnering remarkable momentum in the recent years. The steadily escalating demand due to improving purchasing power is projected to bode well for the global market. QY Research's lates...
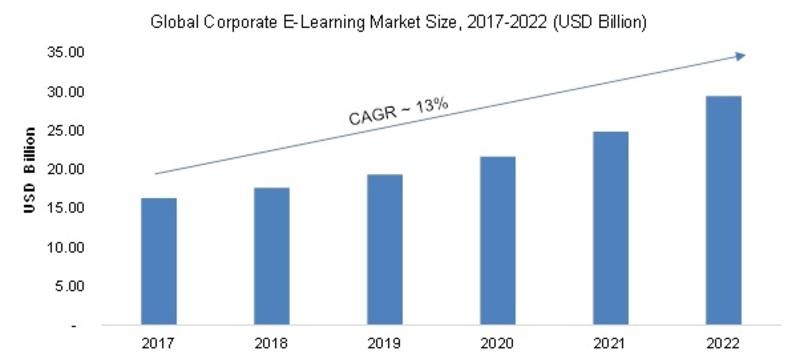
/newsCorporate E-Learning Market - Global Industry Size, Share, Key Players Analysis that are Infor, SkillSoft Corporation, Adrenna, CERTPOINT Systems and others with Regional Forecast to 2022
Overview:
E-Learning is used to enhance the learning procedures for newer job requirements and to make employees sound about the internal and external changes in the market and respective organizations. This method has created considerable differences in the ways of training and developing employee...
